JBRA Assist. Reprod. 2016;20 (4):246-250
REVIEW ARTICLE
doi: 10.5935/1518-0557.20160047
Final Oocyte Maturation in Assisted Reproduction with Human Chorionic Gonadotropin and Gonadotropin-releasing Hormone agonist (Dual Trigger)
1Cenafert - Reproductive Medicine Center, Salvador, BA, Brazil
2São Paulo University – USP, São Paulo, SP, Brazil
3Valencia Infertility Institute – IVI, 14 Ronda del General Mitre, Barcelona, Spain
CONFLICT OF INTERESTS
No conflict of interest have been declared.
ABSTRACT
Final oocyte maturation with Human Chorionic Gonadotropin (hCG) and
ovarian stimulation with Follicle Stimulation Hormone (FSH) combined
with Gonadotrophin-releasing Hormone (GnRH) antagonist to block
Luteinizing hormone (LH) surge is a standard procedure of in vitro
Fertilization (IVF) and Intracytoplasmic Sperm Injection (ICSI).
However, GnRH agonist has been replacing the use of hCG in certain
situations, especially in patients at risk of Ovarian Hyperstimulation
Syndrome (OHSS). Some studies have also shown advantages in the
combined use of GnRH agonist concurrently with hCG in inducing final
oocyte maturation, a treatment known as “Dual Trigger”. In theory, this
method combines the advantages of both induction regimens, and it has
brought promising results. The objective of this study is to compare
Dual Trigger with the use of hCG alone or the use of GnRH agonist
alone. A systematic review of articles on Dual Trigger and a
retrospective cohort study comparing the three methods of induction of
final oocyte maturation have been conducted. It has been found that
Dual Triggering for poor responder patients had a statistically
significant increase in the number of retrieved oocytes, mature
oocytes, and fertilized embryos in the positive beta hCG rate,
implantation rate, and newborn/transferred embryo (TE) rate.
Keywords: Assisted Reproduction, GnRH Agonist Trigger, hCG Trigger, Poor Responder, Immature Oocyte, Ovarian Hyperstimulation Syndrome.
INTRODUCTION
Final oocyte maturation with hCG and ovarian stimulation with FSH
combined with GnRH antagonist to block LH surge is a standard procedure
of in vitro Fertilization (IVF) and Intracytoplasmic Sperm Injection
(ICSI) (Decleer et al., 2014). Human Chorionic Gonadotropin is routinely used for inducing LH surge, thus inducing final oocyte maturation (Schachter et al., 2008). However, the use of hCG can result in Ovarian Hyperstimulation Syndrome (OHSS) (Shapiro et al., 2008). This risk is significantly reduced by replacing hCG with a GnRH agonist (Shapiro et al., 2008; Zilberberg et al., 2015; Lin et al., 2013).
The short half-life of pituitary LH combined with the desensitization
induced by the agonist results in a rapid and irreversible luteolysis,
ideally eliminating the risk of OHSS (Shapiro et al., 2011; Griffin et al., 2012).
In addition to that, some studies have shown that administering GnRH
agonist after the use of GnRH antagonist in an IVF cycle brings about
true benefits for implantation, since the antagonist blocks endometrial
GnRH receptors, worsening endometrial quality. Once the GnRH agonist —
that has a much higher affinity to receptor than the GnRH antagonist —
is administered, a displacement of the antagonist from the receptor
occurs in the endometrium, and it unlocks these receptors, improving
endometrial receptivity (Schachter et al., 2008).
However, as stated earlier, the rapid luteolysis caused by the use of
the agonist consequently leads to an altered luteal phase, and its
final result is the reduction of implantation rates and the increasing
of miscarriage rates, when compared to the use of hCG as a “trigger” (Lin et al., 2013; Griffin et al., 2012).
To solve this problem, some studies have shown that it was possible to
improve implantation rates by administering high doses of progesterone
alone — or combined with estrogen — on the luteal phase after using the
GnRH agonist (Shapiro et al., 2011; Griffin et al., 2012).
Another possibility would be to transfer the vitrification of embryos
into another cycle with more appropriate hormone levels. Furthermore,
in an effort to reduce miscarriage rates, few studies have evaluated
the effects of the use of GnRH agonists associated with hCG 12-35 hours
after triggering with the agonist, and they have shown some
improvements in the luteal phase (Humaidan, 2009). However, the subsequent hCG administration does not act as oocyte maturation (Shapiro et al., 2008).
For approximately eight years, some studies have shown a fourth
possibility: the combination of the use of GnRH agonist concurrently
with hCG to induce final oocyte maturation (Griffin et al., 2012),
a treatment known as “Dual Trigger”, which has been used in patients
with high response, normal response, and poor response, or oocyte
immaturity.
In theory, this method combines the advantages of both induction regimens:
1) it decreases the risk of OHSS by decreasing the dose of hCG;
2) it tends to be a more physiological cycle since there is an FSH peak
addition induced by GnRH agonist generating a larger number of mature
oocytes (Griffin et al., 2014; Haas et al., 2014; Castillo et al., 2013), whereas hCG alone induces LH peak (Decleer et al., 2014);
3) it improves endometrial receptivity for releasing endometrial GnRH receptors;
4) it extends the ovulation time after use of the inductor caused by hCG, also improving the maturation (Zilberberg et al., 2015);
5) there is a better luteal phase recruitment when there is a proven combined use of hCG with GnRH agonist (Shapiro et al., 2011).
However, in practice, there is a statistically significant difference
between the induction of oocyte maturation with isolated hCG, or
isolated GnRH agonist, and the induction of GnRH agonist combined with
hCG in terms of oocyte numbers, embryo quality, and clinical results?
The objectives of this study are:
1) to evaluate “Dual Trigger” studies in high responder patients, poor
responders or patients with oocyte immaturity, and normal responder
patients;
2) to retrospectively evaluate the results obtained with this treatment
in the Valencian Infertility Institute, in Barcelona, Spain.
MATERIAL AND METHODS
Systematic Review
During June 2016, we carried out a systematic review in Pubmed database
using as descriptors (Mesh) the words: “Gonadotropin-Releasing Hormone
/ agonists” AND “Chorionic Gonadotropin, Human”. In total, we found 319
papers. Notwithstanding, we had to discard 306 papers for the following
reasons: 28 were experiments on animals; nearly 170 papers discussed
the use of hCG alone as a “trigger”; 96 argued about the use of GnRH
agonist alone as a “trigger”, and 12 addressed the use of hCG during
the luteal phase, or after the use of GnRH agonist. We ended up with
thirteen papers on the use of Dual Trigger: 6 prospective studies (3
randomized), 6 retrospective cohort studies, and one case report. To
report the results of this systematic review we used the Preferred
Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA)
statement, by Moher et al., 2010.
Retrospective Cohort Study
In addition to the systematic review, a retrospective cohort study was
carried out with data collected from electronic medical records of a
Human Reproduction Clinic in Barcelona, Spain, from June 2014 to March
2015, with high and poor responder patients who had been treated with
Dual Trigger. Dual Triggering has been compared to conventional
treatment (hCG alone, in normal or poor responder patients, and the use
of GnRH agonist in high responders) after controlling ovarian
stimulation with FSH + LH, with doses set based on patient’s weight,
age and history, and the use of GnRH antagonist to block the premature
LH surge. All patients had the luteal phase support with progesterone
(Utrogestan® 1200mg/day, beginning on the day after the ovarian
puncture), and all patients were 40 years old or less, according to the
inclusion criteria. High responder patients were classified according
to the following criteria: more than 20 follicles > 12 mm during
controlled ovarian stimulation, or estradiol > 3000pcg/ml, or risk
factors for OHSS. Among all patients, those who had been treated with
concomitant induction of final oocyte maturation with hCG + GnRH
agonist (Group A: GonasiR 2,500 IU + DecapeptylR 0.2mg) — that is, Dual
Trigger — were compared to the control group, patients who received
only GnRH agonist (Group B: DecapeptylR 0.2mg). Poor responder patients
who classified according to the Bologna criteria were also compared to
patients who had been administered hCG + GnRH agonist (Group C:
OvitrelleR 6,500 IU + DecapeptylR 0.2mg) concurrently with the control
group of patients who had received hCG alone (Group D: OvitrelleR 6,500
IU) as a trigger.
Statistical analysis
Initially, we carried out a
descriptive analysis of the data from which the quantitative variables
were evaluated. In order to assess whether there was a correlation
between quantitative variables, we used the Pearson correlation
coefficient. Some parametric techniques were used in cases where data
normality had been met;
however, nonparametric tests were applied when normality assumption had
been violated. In order to test hypotheses by comparing the averages of
two independent samples, the t-test or the Mann-Whitney U test was
applied.
For data where repeated measures were found for the same variable, we used the nonparametric ANOVA test.
For all tests, we considered a significance level of 5%, and a
confidence interval of 95%. The analyses were performed by means of the
R Core Team (2014) and the Statistical Package for Social Sciences
(SPSS 18, SPSS Inc., Chicago, IL, USA).
Ethics Procedures
Ethics Approval was required
for it is a retrospective study and a systematic review using the
electronic medical records of the Valencian Infertility Institute,
where our research was carried out. The patients did not have their
names disclosed or their behaviors modified. The type of treatment
investigated in this study has already been discussed in other studies,
including prospective and randomized investigations that have shown
positive outcomes.
RESULTS
Systematic Review
Five articles comparing the use of GnRH agonist alone with Dual Trigger
in high responder patients were found: four retrospective cohort study
papers and one prospective study (see Table 1).
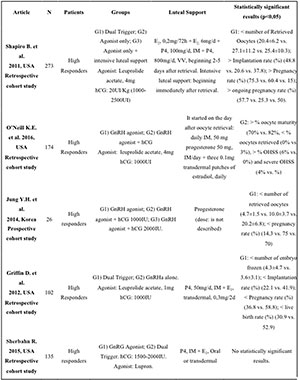
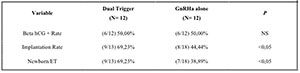
Table 1. Articles on High Responders Patients
The results of these studies were well mixed. While the study by Shapiro et al. (2011)
showed higher implantation and pregnancy rates in the Dual Trigger
group and a lower number of retrieved oocytes and embryo transfers in
this same group, the study by Griffin et al. (2012) showed lower implantation and pregnancy rates in patients who were treated with the Dual Trigger modality. The study by Sherbahn & Catenacci (2015) showed no statistically significant difference between the use of Dual Trigger and isolated GnRH agonist. Jung et al. (2014)
analyzed a small sample size of 26 patients only and found that dual
Triggering with a 2000IU dose of hCG produced a higher pregnancy rate.
The study by O’Neill et al. (2016) showed a higher risk of OHSS in high responder patients who had been treated with the Dual Trigger mode.
Regarding the group of normal responder patients (Table 2),
we found 4 papers comparing the use of hCG alone with the Dual Trigger
mode: 3 prospective and randomized, and one retrospective cohort study.
In this group, the results were quite uniform, and they showed that, by
using the Dual Trigger strategy, there was a higher number of collected
oocytes and mature oocytes, as well as good quality embryos. They have
also shown an increase in implantation and pregnancy rates.
Finally, 3 papers comparing the use of hCG alone with Dual Trigger in
poor responder patients or with immature oocyte and found: 2
prospective, and one retrospective study. In this case, the results
were uniform as well, showing improvements in the number of oocytes,
mature oocytes, fertilized embryos, and good quality embryos, as well
as an increase in the implantation and pregnancy rates using Dual
Trigger (Table 3).
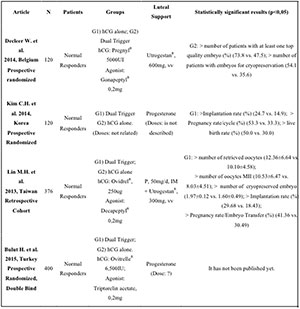
Table 2. Articles on Normal Responder Patients
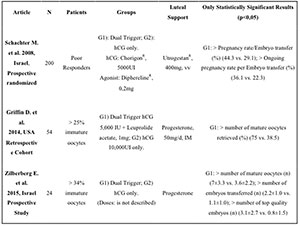
Table 3. Articles on Poor Responders Patients or Patients with Imature Oocytes.
Retrospective cohort study
As described in the methodology section, we designed a retrospective
cohort study, comparing two groups of high responder patients (Dual
Trigger x GnRH agonist alone) and two groups of poor responder patients
(Dual Trigger x hCG alone).
A) High Responder patients
The group of high
responders consisted of a total of 24 patients, 12 in group A (Dual
Trigger) and 12 in B (GnRH alone). There was no statistically
significant difference in terms of age, BMI, basal FSH, AMH, antral
follicle count, stimulation duration, level of estradiol, and
endometrial thickness on the trigger of patients from both groups.
There was no statistically significant difference among collected
oocyte numbers, number of mature oocytes, fertilized embryos, good
quality embryos, transferred embryos, or vitrified embryos.
Group A (Dual Trigger) showed higher implantation and newborn/transferred embryo rates with statistical significance (table 4).
B) Poor Responder Patients
Poor responders were composed of a total of 40 patients divided in two
groups as follows: 18 patients in group C (Dual Trigger) and 22
patients in group D (control group). Comparing both groups, there were
no statistically significant differences in terms of age, BMI, basal
FSH, AMH, antral follicle count, stimulation period, level of
estradiol, and endometrial thickness on the day of trigger between
patients from groups C and D.
Corroborating the results of other studies in the systematic review,
this retrospective study has shown a higher number of oocytes
retrieved, mature oocytes, and embryos fertilized, with statistically
significant difference in patients who were treated with Dual Trigger (Table 5).
There was no difference between the numbers of transferred or vitrified
evolutionary embryos, but group C (Dual Trigger) produced higher
implantation and newborn/ transferred embryo rates.
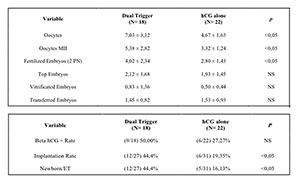
Table 5. Poor responder Patients.
DISCUSSION
This article shows that the use of GnRH agonist combined with hCG in
inducing final oocyte maturation is an excellent alternative after
ovarian stimulation with recombinant FSH and LH, and suppression of
premature LH surge with GnRH antagonist, especially in normal responder
patients, poor responder patients, or patients with immature oocytes.
Our retrospective study and systematic review demonstrated that there
is yet no indication of the use of Dual Triggering mode in high
responder patients, since the results of these studies were
contradictory, and it was impossible to assess the risk of OHSS due to
the low prevalence of this disorder. Supporting the theory explained in
the introduction of this paper, in the cases of normal and poor
responder patients, all reviewed studies — including this retrospective
study — have shown the superiority of treatment with Dual Trigger
regarding the number of mature oocytes, fertilized embryos, and
regarding the implantation rate and the newborn/transferred embryo rate
(Decleer et al., 2014; Schachter et al., 2008; Shapiro et al., 2008; Zilberberg et al., 2015; Lin et al., 2013).
The Dual Trigger strategy significantly improved embryo quality, it
would be interesting to carry out the work in patients with low embryo
quality to assess their actual effectiveness. Furthermore, this method
probably has an advantage in cases of egg donation, to increase the
number of oocytes in normal responder patients. However, we must be
careful, for it is not possible to assess whether Dual Trigger
increases the risk of OHSS when compared to the GnRH agonist alone (Engmann et al., 2008). Moreover, O’Neill et al. (2016) showed that the risk of OHSS has been increasing in high responder patients treated under the Dual Trigger strategy.
It is necessary to compare the use of Dual Trigger with hCG alone in
vitrified embryo cycles followed by vitrified embryo transfers, mainly
because only the effect of embryo quality in pregnancy rates would be
evaluated, and the effects of Dual Trigger or hCG alone would not be
present on the endometrial receptivity. In the papers evaluated, there
was a variation between the dosages of hCG. In the groups of poor
responder patients, from 5,000 IU to 10,000UI, regardless of the dose,
the results were always positive concerning the use of Dual Trigger (Zilberberg et al., 2015; Griffin et al., 2012).
It is worth mentioning that in all the studies, it was necessary to
provide an intensive support during the luteal phase, and some of the
studies did use high doses of progesterone alone, or estrogen and
progesterone in the luteal phase (Decleer et al., 2014).
This undermines the certification, in practice, of the theory that the
Dual Trigger improves the endometrial receptivity, perhaps, this
improvement in implantation rates has been produced by high doses of
progesterone.
The Dual Trigger, to induce oocyte maturation has the
advantage of acting more physiologically as it induces an FSH surge.
The importance of FSH present in the oocyte maturation process has been
proved in a large number of studies that the GnRH agonist increases the
number of mature oocytes (Zeleznik et al., 1974; Richards et al., 1976). Lamb et al. (2011)
carried out a study using an FSH bolus concurrently with the use of hCG
as a trigger, and found an increased number of mature oocytes and
fertilization rate after the use of FSH (19). Our study, as well as the
majority of the studies investigated in this paper, was limited by its
small sample size, requiring, however, a randomized prospective study
with a larger number of patients for more solid conclusions about the
use of Dual Trigger.
In conclusion, according to this retrospective
study, the Dual Trigger used for induction of final oocyte maturation
in normal responders, poor responders, and patients with immature
oocytes, significantly improves the number of collected oocytes, mature
oocytes, and fertilized embryos, as well as improves the beta hCG
positive rate, and the implantation and pregnancy rates. The results
presented in this article reinforce the evidence of improved outcomes
of Human Reproduction treatments using this method.
REFERENCES
Castillo
JC, Moreno J, Dolz M, Bonilla-Musoles F. Successful Pregnancy Following
Dual Triggering Concept (rhCG + GnRH Agonist) in a Patient Showing
Repetitive Immature Oocytes and Empty Follicle Syndrome: Case Report. J
Med Cases. 2013;4:221-6.
Crossref
Decleer
W, Osmanagaoglu K, Seynhave B, Kolibianakis S, Tarlatzis B, Devroey P.
Comparison of hCG triggering versus hCG in combination with a GnRH
agonist: a prospective randomized controlled trial. Facts Views Vis
Obgyn. 2014;6:203-9.
Medline
Engmann
L, DiLuigi A, Schmidt D, Nulsen J, Maier D, Benadiva C. The use of
gonadotropin-releasing hormone (GnRH) agonist to induce oocyte
maturation after co-treatment with GnRH antagonist in high-risk
patients undergoing in vitro fertilization prevents the risk of ovarian
hyperstimulation syndrome: A prospective randomized controlled study.
Fertil Steril. 2008;89:84-91.
Medline Crossref
Griffin
D, Benadiva C, Kummer N, Budinetz T, Nulsen J, Engmann L. Dual trigger
of oocyte maturation with gonadotropin-releasing hormone agonist and
low-dose human chorionic gonadotropin to optimize live birth rates in
high responders. Fertil Steril. 2012;97:1316-20.
Medline Crossref
Griffin
D, Feinn R, Engmann L, Nulsen J, Budinetz T, Benadiva C. Dual trigger
with gonadotropin-releasing hormone agonist and standard dose human
chorionic gonadotropin to improve oocyte maturity rates. Fertil Steril.
2014;102:405-9.
Medline Crossref
Humaidan
P. Luteal Phase rescue in high-risk OHSS patients by GnRHa triggering
in combination with low-dose HCG: a pilot study. Reprod Biomed Online.
2009;18:630-4.
Medline Crossref
Jung
YH, Kim YY, Kim MH, Yoo YJ, Jo JD. Optimal usage of dual trigger to
prevent HSS in a long protocol IVF cycle. Fertil Steril. 2014; 102:e222.
Crossref
Lamb
JD, Shen S, McCulloch C, Jalalian L, Cedars MI, Rosen MP.
Follicle-stimulating hormone administered at the time of human
chorionic gonadotropin trigger improves oocyte developmental competence
in in vitro fertilization cycles: a randomized, double-blinded,
placebo-controlled trial. Fertil Steril. 2011;95:1655-60.
Medline Crossref
Lin
MH, Wu FS, Lee RK, Li SH, Lin SY, Hwu YM. Dual trigger with combination
of gonadotropin-releasing hormone agonist and human chorionic
gonadotropin significantly improves the live-birth rate for normal
responders in GnRH-antagonist cycles. Fertil Steril. 2013;100:1296-302.
Medline Crossref
Moher
D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting
items for systematic reviews and meta-analyses: the PRISMA statement.
Int J Surg. 2010; 8:336-41.
Medline Crossref
O’Neill
KE, Senapati S, Maina I, Gracia C, Dokras A. GnRH agonist with low-dose
hCG (dual trigger) is associated with higher risk of severe ovarian
hyperstimulation syndrome compared to GnRH agonist alone. J Assist
Reprod Genet. 2016;33:1175-84.
Medline Crossref
Richards
JS, Ireland JJ, Rao MC, Bernath GA, Midgley AR Jr, Reichert LE Jr.
Ovarian follicular development in the rat: hormone receptor regulation
by estradiol, follicle stimulating hormone and luteinizing hormone.
Endocrinology. 1976;99:1562-70.
Medline Crossref
Schachter
M, Friedler S, Ron-El R, Zimmerman AL, Strassburger D, Bern O, Raziel
A. Can pregnancy rate be improved in gonadotropin-releasing hormone
(GnRH) antagonist cycles by administering GnRH agonist before oocyte
retrieval? A prospective, randomized study. Fertil Steril.
2008;90:1087-93.
Medline Crossref
Shapiro
BS, Daneshmand ST, Garner FC, Aguirre M, Thomas S.
Gonadotropin-releasing hormone agonist combined with a reduced dose of
human chorionic gonadotropin for final oocyte maturation in fresh
autologous cycles of in vitro fertilization. Fertil Steril.
2008;90:231-3.
Medline Crossref
Shapiro
BS, Daneshmand ST, Garner FC, Aguirre M, Hudson C. Comparison of
‘‘triggers’’ using leuprolide acetate alone or in combination with
low-dose human chorionic gonadotropin. Fertil Steril. 2011; 95:2715-7.
Medline Crossref
Sherbahn
R, Catenacci M. High Live Birth Rates in IVF High Responders Using
Either a Lupron Trigger Alone (agonist trigger) or Using a Dual Trigger
if Intensive Luteal Support is Given. Fertil Steril. 2014;102:e316.
Crossref
Zeleznik
AJ, Midgley AR, Reichert LE Jr. Granulosa cell maturation in the rat:
Increased binding of human chorionic gonadotropin following treatment
with follicle stimulating hormone in vitro. Endocrinology.
1974;95:818-25.
Medline Crossref
Zilberberg
E, Haas J, Dar S, Kedem A, Machtinger R, Orvieto R. Co-administration
of GnRH-agonist and hCG, for final oocyte maturation (double trigger),
in patients with low proportion of mature oocytes. Gynecol Endocrinol.
2015; 31:145-7.
Medline Crossref