JBRA Assist. Reprod. 2019;23(3):268-272
REVIEW
doi: 10.5935/1518-0557.20190018
Pregnancy rates from natural and artificial cycles of women submitted to frozen embryo transfers: a metanalysis
1Laboratorio de Reprodução Humana (LabRep) / FM / HC - Federal University of Goiás - Brazil
2Pontifical Catholic University of Goiás - Brazil
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest
ABSTRACT
Objective: To compare pregnancy rates from natural and artificial cycles of women submitted to frozen embryo transfers.
Methods: A systematic review was performed by PubMed search using the following algorithm: (endometrial [All Fields] AND preparation [All Fields]) AND (("freezing"[MeSH Terms] OR "freezing"[All Fields] OR "frozen"[All Fields]) AND thawed [All Fields]) and (natural cycles) AND (artificial cycle). Inclusion criteria: prospective and retrospective cohort studies. Exclusion criteria: use of hCG in the natural cycle, oocyte donors, and use of disused freezing techniques. Data were analyzed with the SPSS v.23 software and with a significance level of 5%. The meta-analysis was performed with RevMan 5.3 software. I² was calculated.
Results: 709 papers were retrieved. Five studies fulfilled the inclusion and exclusion criteria. From these studies, we analyzed 8,968 natural or artificial cycles. A contingency table compared the results of the natural and artificial cycles and the number of clinical pregnancies obtained in each selected paper. The I² test resulted in high statistical heterogeneity (I²=77%). Studies by Morozov et al. (2007) and Zheng et al. (2015) obtained statistically significant results (p<0.03 and p<0.001): Morozov et al. (2007) found a higher pregnancy rate within natural cycles, and Zheng et al. (2015) found more positive outcomes when analyzing artificial cycles. The remaining selected studies did not show any statistical significance.
Conclusion: There is insufficient scientific evidence to state that the artificial cycle yields better pregnancy rates than the natural cycle in women submitted to frozen embryo transfer. Limitations of this study include a small number of papers and heterogeneity among the studies.
Keywords: endometrium, pregnancy rate, cryopreservation
INTRODUCTION
Adequate endometrial hormonal preparation is fundamental for the success of frozen embryo transfers in vitro fertilization cycles (Harper, 1992). Endometrium preparation can be achieved in a natural cycle after spontaneous ovulation or after using exogenous estrogen and progesterone.
In the natural ovulatory cycle, the hormonal secretion of the ovaries controls endometrium development. It undergoes a series of foreseeable changes associated with follicular development, ovulation and corpus luteum (Psychoyos, 1973). In the artificial cycle, estrogen and progesterone supplementation is used to mimic the normal cycle. The goal is to achieve adequate endometrial thickness to receive the embryo (Paulson, 2011).
Cryopreservation enables embryo transfer after ovarian stimulation for oocyte collection. It makes it possible to avoid ovarian hyperstimulation syndrome and to plan the options for the moment of the transfer (Corbett et al., 2014). The frozen embryo transfer success is closely linked to the exact synchronization between endometrial maturation and embryo development, as well as the transfer technique used (Diedrich et al., 2007; Mains & Van Voorhis, 2010). Reviews comparing the several endometrial preparation techniques were inconclusive (Glujovsky et al., 2010; Groenewoud et al., 2013; Ghobara et al., 2017).
The objective is to compare pregnancy rates among natural and artificial cycles of women submitted to frozen embryo transfers.
MATERIALS AND METHODS
A meta-analysis was performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statements, with an active search in the PubMed database from 2016-2017. The following descriptors were used: "Cryopreservation", "Embryo Transfer", "Endometrium/Estrogens", "Female", "Fertility Agents", "Pregnancy", "Pregnancy Rate".
The search algorithm used in the PubMed platform was the following: (Endometrial [All Fields] AND preparation [All Fields]) AND (("freezing" [MeSH Terms] OR "freezing" [All Fields] OR "frozen" [All Fields]) AND thawed [All Fields]) and (natural cycles) AND (artificial cycle).
Two independent reviewers evaluated the available papers. In case of disagreement, a third reviewer was called upon for further assessment. The inclusion criteria were description of endometrial preparation regimens, prospective and retrospective cohort studies comparing artificial cycles and natural cycles. The exclusion criteria were studies that lack information about endometrial preparation regimens, those that have not compared natural and artificial cycles, and those that did not describe the clinical pregnancy rates. Studies using hCG in the natural cycle (natural cycle modified) and those using oocyte donors were excluded.
In the pre-selection phase, we found 709 studies with the application of the descriptors. After using the inclusion and exclusion criteria, papers with over 15 years of publication were also excluded, because the most recent embryo freezing techniques are superior. A flow diagram demonstrating the process of paper selection and eligibility is demonstrated in Figure 1. Five retrospective cohort studies fulfilled the eligibility criteria and were selected.
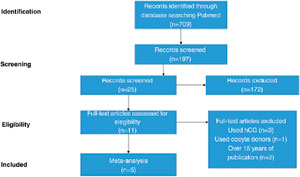
Figure 1. Flow diagram of the paper selection flowchart
The data was analyzed with the aid of the statistical package SPSS version 23, adopting a level of significance of 5% (p<0.05). The association between the type of gestation and the positive or negative outcome was performed based on the Pearson Chi-square test.
The meta-analysis was performed using the RevMan 5.3 software. The I2 was calculated for statistical heterogeneity. Values higher than 30% indicate heterogeneity. A random effect model was used in case of heterogeneity and a fixed-effect model in its absence. The Haenszel method was applied to estimate the pooled effects sizes. We used a significance level of p less than 0.05.
RESULTS
A total of five studies were included with 8,968 natural or artificial cycles investigated. Table 1 depicts the variables extracted from the studies, including outcomes and protocols. This table includes the results of the natural and artificial cycles, and the number of clinical pregnancy rates obtained in each selected paper were compared.
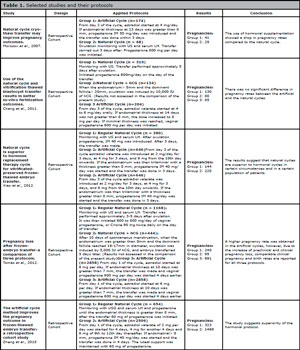
Table 1. Selected studies and their protocols
The I2 test was applied to look for heterogeneity. Given the high statistical heterogeneity (I2=77%), we applied the random effects model to the meta-analysis.
Table 2 shows an association between the type of pregnancy and the outcome from each paper.
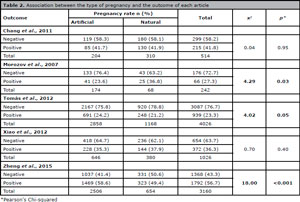
Table 2. Association between the type of pregnancy and the outcome of each article
Figure 2 presents a Forest Plot for the results obtained in the selected papers, with the comparison between artificial cycles versus natural cycles. We found no significant difference for the different types of stimulation protocol and pregnancy outcomes.
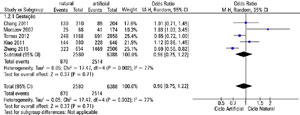
Figure 2. Artificial cycle x Natural cycle forest plot
In the studies analyzed, only Morozov et al. (2007) and Zheng et al. (2015) found statistically significant results, being favorable for opposite results. While the former had a higher natural-cycle pregnancy rate, the latter had more positive outcomes with the artificial cycle. The other selected studies did not show statistical significance to support one cycle modality over the other.
DISCUSSION
The estrogen administration doses and protocols varied in the interventions of the artificial cycles. Two studies used increasing doses of estrogen: Xiao et al. (2012) and Zheng et al. (2015). The other papers used constant estrogen doses. Studies suggest that the management of these doses may influence the final outcome, showing that using constant doses can result in a higher pregnancy rate (Madero et al., 2016). Studies were excluded where oocyte donations were used, since it is known that this is an independent factor that positively alters the results. Studies that select only specific types of infertility causes could further elucidate the issue.
Morozov et al. (2007) obtained significantly better results with the natural cycle and discuss the benefits of not using hormones, primarily in women with regular menstrual cycles. Some experimental models have shown that the implantation window would be smaller when the endometrium is exposed to higher levels of estrogen (Ma et al., 2003). The endometrial thickness of patients who had natural cycle gestation showed an average value close to 10 mm, corroborating that the average levels of the hormone present in the natural cycle would be sufficient to provide a developed endometrium, a predictor of success (Zhang et al., 2005), as well as a larger window for embryo implantation. Chang et al. (2011) argue that patients with normal ovarian function would benefit more from natural cycles since supraphysiological concentrations of estrogen would reduce the expression of the β3-subunit integrin and leukemia inhibitory factor (LIF) in the endometrium (Chen et al., 2008). The correlation between pregnancy rates and endometrial thickness was analyzed, and there was a statistically significant difference showing a good relationship between larger sizes and the success of the process.
Xiao et al. (2012) argue that a certain bias may have influenced their study, since it is retrospective and the patients had the method chosen according to their menstrual history - they were not randomized. They point out that in the artificial cycle there may be suppression of several hormones essential to maintain the pregnancy. Tomás et al. (2012) showed a higher number of pregnancies with artificial cycles, but also higher rates of abortion. The transfer of frozen embryos has higher loss rates when compared to the transfer of fresh embryos (Farr et al., 2007), without distinction of the protocol used. It is argued that there was a greater number of patients with polycystic ovary syndrome submitted to an artificial cycle, which may have influenced gestational loss. Similarly, Zheng et al. (2015) showed higher pregnancy rates in artificial cycles, but unlike the previous study, abortion rates were similar between the groups. The authors suggest that this happens not only because of the patient’s characteristics, but also because of the transfer method used in their study, in the blastocyst stage. They reported that hormonal supplementation is simpler and more flexible in relation to the moment of transfer, and it is better for women with irregular menstrual cycles.
This study presents some limitations that should be considered as they may jeopardize data generalization. These include the small number of studies retrieved and the high heterogeneity rate found among them. The lack of evaluation of additional variables such as patient’s age and embryo quality is also a limitation. However, we demonstrated that there is an important gap in the literature concerning this topic, as controversial results are found. In addition, more rigorous definitions and data access standardization should be considered in order to favor comparability among studies.
A method may be more favorable for a given population and such individualities could not be measured in the analysis we carried out. It is believed that other factors should be taken into account in choosing the method, such as costs, side effects and ease of use by the patient. Further randomized prospective studies are needed to define the best course of action to be followed, with greater efficiency, cost-effectiveness, safety and convenience.
CONCLUSION
There is insufficient evidence to state that the artificial cycle offers better pregnancy rates than the natural cycle in women undergoing frozen embryo transfers. The results of this review should be carefully considered because of the small number of studies and the heterogeneity between the studies. It is suggested that more prospective studies comparing natural and artificial cycles are performed.
This study was presented at the XXII Brazilian Congress of Assisted Reproduction
REFERENCES
Chang EM, Han JE, Kim YS, Lyu SW, Lee WS, Yoon TK. Use of the natural cycle and vitrification thawed blastocyst transfer results in better in-vitro fertilization outcomes: cycle regimens of vitrification thawed blastocyst transfer. J Assist Reprod Genet. 2011;28:369-74.
Medline Crossref
Chen QJ, Sun XX, Li L, Gao XH, Gemzell-Danielsson K, Cheng LN. Effects of ovarian stimulation on endometrial integrin beta3 and leukemia inhibitory factor expression in the peri-implantation phase. Fertil Steril. 2008;89:1357-63.
Medline Crossref
Corbett S, Shmorgun D, Claman P; Reproductive Endocrinology Infertility Committee; Special Contributor. The Prevention of Ovarian Hyperstimulation Syndrome. J Obstet Gynaecol Can. 2014;36:1024-33.
Medline Crossref
Diedrich K, Fauser BC, Devroey P, Griesinger G; Evian Annual Reproduction (EVAR) Workshop Group. The role of the endometrium and embryo in human implantation. Hum Reprod Update. 2007;13:365-77.
Medline Crossref
Farr SL, Schieve LA, Jamieson DJ. Pregnancy loss among pregnancies conceived through assisted reproductive technology, United States, 1999-2002. Am J Epidemiol. 2007;165:1380-8.
Medline Crossref
Ghobara T, Gelbaya TA, Ayeleke RO. Cycle regimens for frozen-thawed embryo transfer. Cochrane Database Syst Rev. 2017;7:CD003414.
Medline Crossref
Glujovsky D, Pesce R, Fiszbajn G, Sueldo C, Hart RJ, Ciapponi A. Endometrial preparation for women undergoing embryo transfer with frozen embryos or embryos derived from donor oocytes. Cochrane Database Syst Rev . 2010;1:CD006359.
Medline Crossref
Groenewoud ER, Cantineau AE, Kollen BJ, Macklon NS, Cohlen BJ. What is the optimal means of preparing the endometrium in frozen-thawed embryo transfer cycles? A systematic review and meta-analysis. Hum Reprod Update. 2013;19:458-70.
Medline Crossref
Harper MJ. The implantation window. Baillieres Clin Obstet Gynaecol. 1992;6:351-71.
Crossref
Ma WG, Song H, Das SK, Paria BC, Dey SK. Estrogen is a critical determinant that specifies the duration of the window of uterine receptivity for implantation. Proc Natl Acad Sci U S A. 2003;100:2963-8.
Medline Crossref
Madero S, Rodriguez A, Vassena R, Vernaeve V. Endometrial preparation: effect of estrogen dose and administration route on reproductive outcomes in oocyte donation cycles with fresh embryo transfer. Hum Reprod. 2016;31:1755-64.
Medline Crossref
Mains L, Van Voorhis BJ. Optimizing the technique of embryo transfer. Fertil Steril. 2010;94:785-90.
Medline Crossref
Morozov V, Ruman J, Kenigsberg D, Moodie G, Brenner S. Natural cycle cryo-thaw transfer may improve pregnancy outcome. J Assist Reprod Genet. 2007;24:119-23.
Medline Crossref
Paulson RJ. Hormonal induction of endometrial receptivity. Fertil Steril. 2011;96:530-5.
Medline Crossref
Psychoyos A. Hormonal control of ovoimplantation. Vitam Horm. 1973;31:201-56.
Medline Crossref
Tomás C, Alsbjerg B, Martikainen H, Humaidan P. Pregnancy loss after frozen-embryo transfer--a comparison of three protocols. Fertil Steril. 2012;98:1165-9.
Medline Crossref
Xiao Z, Zhou X, Xu W, Yang J, Xie Q. Natural cycle is superior to hormone replacement therapy cycle for vitrificated-preserved frozen-thawed embryo transfer. Syst Biol Reprod Med. 2012;58:107-12.
Medline Crossref
Zhang X, Chen CH, Confino E, Barnes R, Milad M, Kazer RR. Increased endometrial thickness is associated with improved treatment outcome for selected patients undergoing in vitro fertilization-embryo transfer. Fertil Steril. 2005;83:336-40.
Medline Crossref
Zheng Y, Dong X, Huang B, Zhang H, Ai J. The artificial cycle method improves the pregnancy outcome in frozen-thawed embryo transfer: a retrospective cohort study. Gynecol Endocrinol. 2015;31:70-4.
Medline Crossref